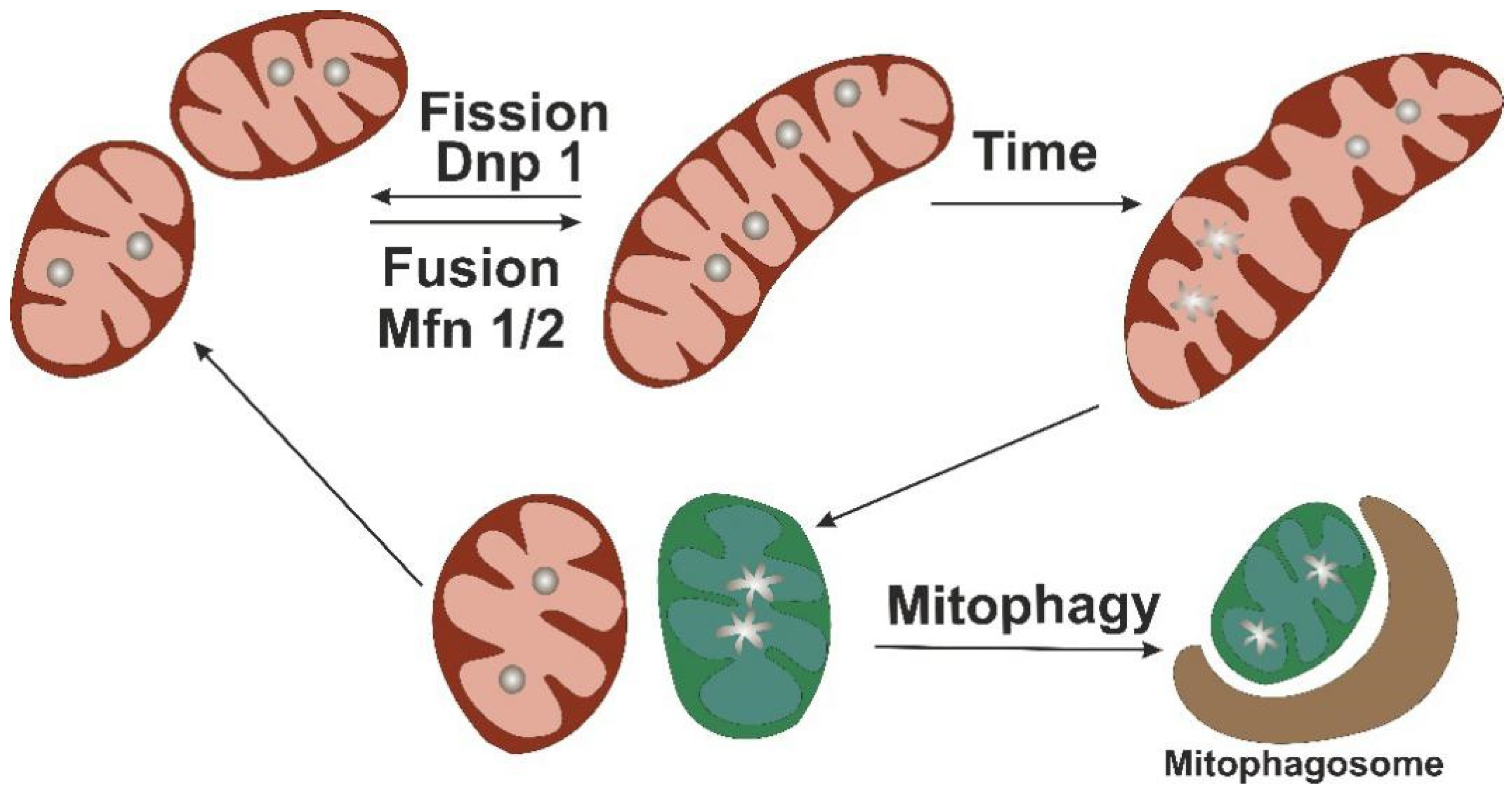
6 However, no selective Met inhibitors, such as SU11274 and Tepotinib, have been proven despite the encouraging wave of recent drug approvals for HCC. Currently, at least 17 Met inhibitors, including JNJ-38877605, GEN-203, and ARQ-197, are undergoing clinical trials. Various cytoplasmic effectors, including PI3K, Ras, PLC-γ, Shc, and SHP2, are recruited to the docking site and subsequently activated. 2, 3, 4, 5 Upon activation, the tyrosine residues Y1234 and Y1235 in the kinase domain of Met are phosphorylated, which leads to auto-phosphorylation of the C-terminal multi-substrate docking site, Y1349, and Y1356. 1 High hepatocyte growth factor (HGF) levels in serum or overexpression of Met in hepatocellular carcinoma (HCC) are closely associated with early recurrence, 2 and patients with high expression levels of Met usually have low 5-year survival rates after curative surgical resection. Liver cancer is the second leading cause of cancer death worldwide because of the high rate of metastasis. These findings reveal a novel and noncanonical pathway of Met receptor tyrosine kinase in the regulation of mitochondrial activities, which may provide a therapeutic target for metastatic HCC. Fragmented mitochondria fueled actin filament remodeling and lamellipodia or invadopodia formation to facilitate cell metastasis in hepatocellular carcinoma (HCC) cells both in vitro and in vivo. Fis1 pY38 promoted mitochondrial fission by recruiting the mitochondrial fission GTPase dynamin-related protein-1 (Drp1) to mitochondria. Mechanically, we found that Met directly phosphorylated outer mitochondrial membrane protein Fis1 at Tyr38 (Fis1 pY38). The contacts between activated Met kinase and mitochondria formed dramatically, and an intact HGF/Met axis was necessary for dysregulated mitochondrial fission and cancer cell movements. Here, using structured illumination microscopy (SIM) and high spatial and temporal resolution live cell imaging, we identified mitochondrial trafficking of receptor tyrosine kinase Met. Dysregulated mitochondrial dynamics are responsible for the progression and metastasis of many cancers. Mitochondria are highly dynamic and undergo fission and fusion to maintain a functional mitochondrial network. All rights reserved.Met tyrosine kinase, a receptor for a hepatocyte growth factor (HGF), plays a critical role in tumor growth, metastasis, and drug resistance. We suggest that deeper understanding of the regulatory mechanisms within this system and downstream implications could benefit in understanding and intervention of these conditions.Īge-related disease Aging Fission Fusion Mitochondrial dynamics.Ĭopyright © 2020 The Author(s). Finally, we discuss the current evidence implicating these processes in age-related human pathologies, such as neurodegenerative or cardio-metabolic diseases. Here, we discuss the mechanisms of mitochondrial fission and fusion, the current evidence of their role in aging of multicellular organisms, and how these connect to cell cycle regulation, quality control, and transmission of energy status. There does exist, however, a large body of evidence connecting mitochondrial dynamics to other aging-related cellular processes and implicates them in a number of human diseases. While many mitochondrial processes are already characterized in relation to aging, specific evidence in multicellular organisms causally linking mitochondrial dynamics to the regulation of lifespan is limited. Mitochondria separate and merge using fission and fusion processes in response to changes in energy and stress status. Crucial to mitochondrial regulation is the dynamic nature of their network structure. Dysregulation of mitochondrial function is one of the classical hallmarks of aging, and mitochondrial interventions have repeatedly been shown to improve outcomes in age-related diseases. The mitochondria is the major hub to convert energy for cellular processes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
